Advancing Genetics Care Together
Your Partner in Delivering Actionable Genomic Medicine to More Patients
We are genomic and genetic experts who partner with healthcare organizations like yours to deliver best-in-class genetic counseling and services to more patients, bringing the promise of precision medicine to more lives. Expand your genetics programs with our telehealth services and national network of clinical genomic experts.

Helping You Deliver
Better Care



Helping Patients Navigate the Genetic Counseling Landscape
Genetic counseling and services are transforming healthcare by making care more targeted, personalized and actionable. Building or expanding your genetics programs should be a seamless experience—for you and your patients.
We Are Genetics Experts Helping You Care for Your Patients
Through our nationwide virtual care delivery model, we provide genetics care to your patients when and where it is most convenient. We have an extensive team of clinicians, including medical geneticists, genetic counselors, physicians and pharmacists who specialize in genetic medicine.
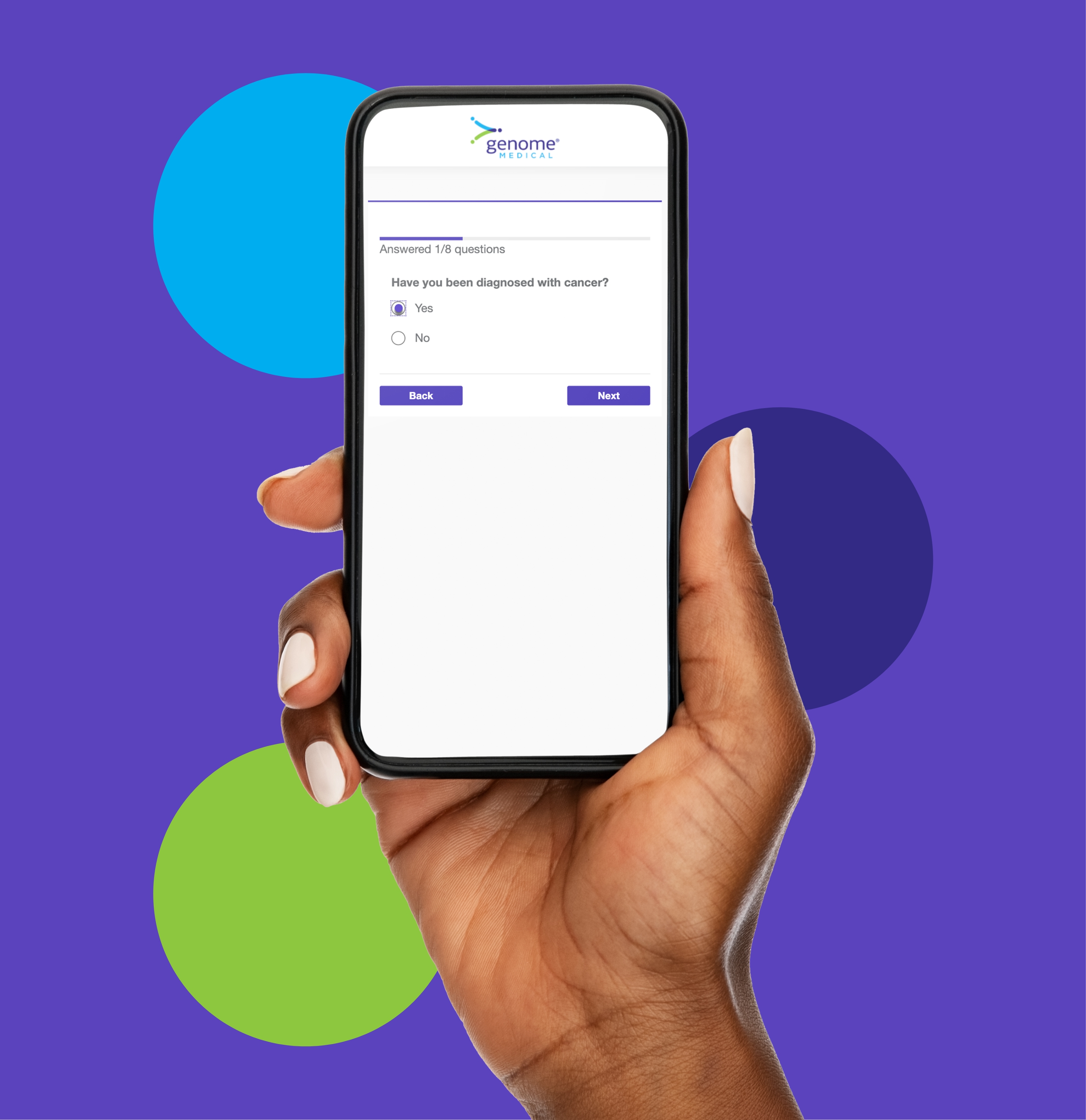
A Seamless Genetic Counseling and Services Experience
Your patients can speak with our genetics experts right from home. Our genetic counselors help your patients understand their genetic risk so they can make informed health care decisions.
Who We Support
We partner with hospitals and health systems, biopharmaceutical organizations and genetic testing labs to help them reach more patients more efficiently and make genetic testing actionable to improve patient care.
-
Hospitals & Health Systems
From large multi-site systems to smaller community hospitals, we help you reach more patients.
-
Biopharmaceutical Organizations
Whether it's support for clinical trials to product launch or market expansion, we help you more efficiently identify and understand your patient population.
-
Genetic Testing Labs
We support your testing services with genetic counseling for patients and help facilitate ordering if medically necessary.
Putting Patients First



